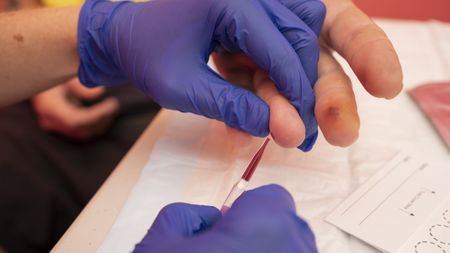
A recent Australian trial revealed that a fingerstick test for hepatitis B DNA is as precise as standard lab testing. This breakthrough could lead to broader accessibility in remote and resource-limited areas. The study, published in the Journal of Clinical Microbiology, demonstrated that this point-of-care test can provide results within an hour and can be conducted in decentralized clinics.
The fingerstick test addresses delays associated with traditional laboratory testing. The Kirby Institute at the University of New South Wales in Australia emphasized the significance of this advancement. Professor Gail Matthews, who spearheaded the research, highlighted the test’s high accuracy, comparable to conventional methods.
This development has the potential to enhance global access to testing and treatment, particularly in regions with limited testing facilities. Hepatitis B, a liver-targeting viral infection, impacts around 254 million individuals globally and leads to over 1 million deaths annually. Despite being preventable through vaccination, only a small percentage of those with chronic hepatitis B receive treatment, according to the World Health Organization (WHO).
The current standard for hepatitis B DNA testing necessitates venous blood samples processed in centralized labs, causing inconvenience for patients who may have to travel long distances and endure prolonged wait times for results. In contrast, the new point-of-care test can be conducted in small health clinics using a finger stick blood sample. This method, delivering results within 60 minutes, can be administered by a wider range of healthcare workers.
While fingerstick testing has been effective for various infectious diseases like hepatitis C, its suitability for hepatitis B DNA was previously unknown. The researchers believe that this innovative test could aid in achieving the WHO’s goal of eliminating hepatitis B as a public health threat by 2030.